Building a Microbial Army
Natural bacteria picked up by honeybees from flowers while collecting nectar and pollen reside predominantly in honey bee midgut and hindgut. Gut bacteria naturally found in honey bees are dynamic. During development of the larvae, bacterial population fluctuates. Larvae receive some bacteria from the nurse bees feeding them. During pupation, the gut lining is shed, and the gut of a newly emerging adult honeybee is sterile. The gut is quickly repopulated with characteristic microbiota. How does this happen? Main pathways are oral trophallaxis, interaction with hive material, and fecal-oral transmission.
Microbial Soldiers
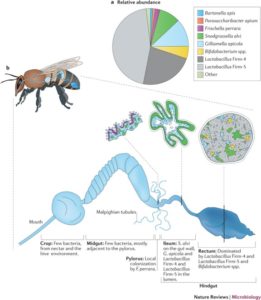
Figure 1. Microbial composition in honeybee gut is dominated by lactic acid producing bacteria to aid in immune response and digestion (12).
The intestinal tract of honey bees contains diverse microbial lifeforms. All Apis species possess a similar gut microbiota, major constituents of which are beneficial Lactic Acid Bacteria (LAB), bacteria such as Lactobacillus and Bifidobacterium (Figure 1). Normal, healthy microflora provides antimicrobial substances to protect from pathogens and digest complex carbohydrates in the bee diet. Maintaining healthy populations of beneficial bacteria in the honey bee gut is therefore vital to hive resiliency and longevity.
When Good Prevails
LAB and other organic acid producing bacteria modulate the honey bee host gut environment to deter pathogens using a variety of mechanisms:
-Competing for nutrients and gut epithelial adhesion
-Acidifying the honey bee gut to deter acid-sensitive pathogenic bacteria
-Activating innate immune responses via communication pathways between microbes and host cells (8).
Abundant members of the honey bee gut microbiome are well characterized for inhibiting growth of other bacteria in mixed culture (3,5,17). LAB increase intestinal mucin production in the gut and thus inhibit pathogen adherence. When pathogens are unable to adhere to gut epithelium, they are more easily eliminated from the host (20). It has been shown that LAB reduce honey bee mortality from diseases such as European Foulbrood caused by the bacteria Melissococcus plutonius and American Foulbrood caused by the bacteria Paenibacillus larvae (1,7,10,22,23). These pathogens increase in the absence of the beneficial protective bacteria. Presence of LAB also decreases pathogens associated with the parasitic Varroa destructor mites and Nosema ceranae (3,4,14).
When Evil Prevails
The hallmark of a normal microbiome is symbiotic cooperation that is the opposite of, and prevents, an infection. It is a homeostatic – self-regulating – ecosystem, or community. Yet nothing lasts forever. The abundance and naturally found balance of bacteria in the gut can be disrupted. Antibiotics, stress, disease disrupt the balance of bacteria in the gut. This results in a condition called dysbiosis.
Honey bee colonies have declined rapidly from 6 million in the 1940s to about 2.6 million today (2). High annual honeybee colony loss is still observed and has become the norm for beekeepers. Gut health plays a significant role in innate host immune response and adaptability to the multitude of stressors honey bees face today.
Factors influencing microbial gut imbalance and worldwide decline in honey bee populations include dependence on pesticides and herbicides for large scale commercial agriculture, decreased biodiversity, quality and quantity of foraging resources, and prophylactic use of antibiotics leading to resistance.
In 2016, antibiotic use in honey bees became regulated and now requires veterinary directive. Despite regulated antibiotics, overuse persists in the U.S. beekeeping industry today. Antibiotic exposure perturbs the individual Apis mellifera (honey bee) gut microbiota, resulting in dysbiosis, weight loss, and death (16,18).
In addition to direct use of antibiotics, honey bee foragers are exposed to high levels of fungicides and pesticides with antimicrobial activity which present a serious threat to bees’ protective gut bacteria. Not only individual foragers, but the whole honey bee colony is exposed to environmental pesticides, since pesticide levels are detectable throughout the year in all hive products, from wax to pollen to honey (16).
Honey bee enzymatic detoxification pathways are often compromised by the synergistic effects of multiple pesticides, lowering the lethal dose threshold (19). One pesticide with documented antimicrobial activity is glyphosate. Glyphosate is a strong antibiotic and toxic to microorganisms including Lactobacillus spp. and Bifidobacterium spp. (6,8,10,11,12,20). Additionally, the antimicrobial effects associated with glyphosate can increase virulence of fungal pathogens (9,11,12).
Video interview with Dr. Don Huber discussing effects of Glyphosate on honey bee health.
Turns out, loss of these important and dominant inhabitants of healthy honeybee gut has detrimental consequences for bee health. In 2007 study, “A metagenomic survey of microbes in honey bee colony collapse disorder” Cox-Foster and colleagues found decreased levels of Lactobacillus and Bifidobacterium in honey bee colony collapse disorder.
Strength in Microbial Numbers
No standardized testing of honey bee microbial levels on a nationwide survey currently exists. Strong Microbials Inc. in Milwaukee, WI cultures LAB from the intestinal tract of the bees on MRS (de Man, Rogosa, and Sharpe), and provides diagnostic evaluation of honey bee microbial levels to customers in the U.S. Analysis of honey bee samples sent to Strong Microbials over the 2012-2017 years indicates a great temporal but not geographic variation in the abundance of the LAB in honey bee gut. This seasonal variation in LAB levels is shown below:

Figure 2. Seasonal variation in lactic acid bacteria abundance in honey bee samples over time. In 2017, the average monthly temperatures in the contiguous United States as recorded by NOAA was 70.3°F (June), 75.7°F (July), 72.7°F (August), 66.3°F (September). Data indicates that LAB concentration in the gut is not stable and fluctuates seasonally. Peaks during June, rising again in September may indicate microbial growth during times of sugar-feeding or increased forage. Supplementation of probiotics is necessary to restore levels of LAB throughout the season.
Deploy the Microbial Army
There is a solution: probiotics. Strong Microbials developed SuperDFM-HoneyBee, a first probiotic supplement for honeybees that contains several commensal strains of Bacillus, Lactobacillus and Bifidobacterium, nutritional yeast and digestive enzymes.
SuperDFM-HoneyBee probiotic supplement restores LAB constituents of the honey bee microbiome. Administering LAB as freeze-dried cultures is recommended as a probiotic regimen for bees as it preserves viability and stability of the product. Colonies that received SuperDFM-HoneyBee have increased LAB levels compared to control bee hives, are stronger and healthier, with lower incidence of Nosema, lower virus incidence, and better winter survival rates.
Peer reviewed publications unequivocally demonstrate that probiotic strains Lactobacillus and Bacillus inhibit growth of bacterial pathogens such as Paenibacillus larvae that causes American foulbrood disease Forsgren, E, et. al. (2010), Melissococcus plutonius that causes European foulbrood disease Wu M., et. al (2014), and fungal pathogen Ascophaera apis that causes chalkbrood Sabaté D.C., et.al. (2009). Unlike antibiotics, probiotics can be used throughout the year and do not leave any residue in the honey or hive products.
Prophylactic supplementation of probiotics results in statistically significant (P <0.001) decreases in the total number of potentially pathogenic microbes while increasing the intestinal colonization of beneficial bacteria (17). Probiotics promote host defenses by encouraging beneficial bacteria to create an unfavorable environment for pathogenic bacteria to establish residency in the honey bee gut.
References:
- Alippi A, Reynaldi F. 2006. Inhibition of the growth of Paenibacillus larvae, the causal agent of American foulbrood of honeybees, by selected strains of aerobic spore-forming bacteria isolated from apiarian sources. Journal of Invertebrate Pathology 91(3): 141-6; doi:10.1016/j.jip.2005.12.002
- ARS Honey Bee Health and Colony Collapse Disorder. 2018. Retrieved from https://www.ars.usda.gov/oc/br/ccd/index/#labs
- Audisio, M, Sabate, D, Benitez-Ahrendts, M. 2015. Effect of Lactobacillus johnsonii CRL1647 on different parameters of honeybee colonies and bacterial populations of the bee gut. Beneficial Microbes: 6(5): 687-95; doi:10.3920/BM2014.0155
- Baffoni L, Gaggìa F, Alberoni D, Cabbri R, Nanetti A, Biavati B, Di Gioia D. 2015. Effect of dietary supplementation of Bifidobacterium and Lactobacillus strains in Apis mellifera L. against Nosema ceranae. Beneficial microbes 7: 1-8; doi:10.3920/BM2015.0085
- Belhadj H, Harzallah D, Bouamra D, Khennouf S, Dahamna S, Ghadbane M. 2014. Phenotypic and Genotypic Characterization of Some Lactic Acid Bacteria Isolated from Bee Pollen: A Preliminary Study. Bioscience of Microbiota, Food and Health 33(1): 11-23; doi:10.12938/bmfh.33.11
- Clair E, Linn L, Travert C, Amiel C, Séralini G, Panoff J. 2012. Effects of Roundup(®) and glyphosate on three food microorganisms: Geotrichum candidum, Lactococcus lactis subsp. cremoris and Lactobacillus delbrueckii subsp. bulgaricus. Current Microbiology 64(5): 486-91
- Evans J, Armstrong T. 2006. Antagonistic interactions between honey bee bacterial symbionts and implications for disease. BMC Ecology 6:4; doi:10.1186/1472-6785-6-4
- Hamdi C, Balloi A, Essanaa J, Crotti E, Gonella E, Raddadi N, Ricci I, Boudabous A, Borin S, Manino A, Bandi C, Alma A, Daffonchio D, Cherif A. 2010. Gut microbiome dysbiosis and honeybee health. Journal of Applied Entomology 135(7): 524-533
- Johal G, Huber D, Glyphosate effects on diseases of plants. 2009. European Journal of Agronomy 31(3):144-152
- Killer J, Dubna S, Sedlacek I, Svec P. 2014. Lactobacillus apis nov., from the stomach of honeybees (Apis mellifera), having an in vitro inhibitory effect on the causative agents of American and European foulbrood. International Journal of Systematic and Evolutionary Microbiology 64: 152-157: doi:10.1099/ijs.0.053033-0
- Kremer R, Means N. 2009. Glyphosate and glyphosate-resistant crop interactions with rhizosphere microorganisms. European Journal of Agronomy 31(3):153-161
- Krüger M, Shehata A, Schrödl W, Rodloff A. 2013. Glyphosate suppresses the antagonistic effect of Enterococcus spp. on Clostridium botulinum. Anaerobe 20:74-8
- Kwong W, Moran N. 2016. Gut microbial communities of social bees. Nature Reviews. Microbiology 14(6):374-84; doi:10.1038/nrmicro.2016.43
- Maggi M, Negri P, Plischuk S, Szawarski N, Piano F, Feudis L, Eguaras M, Audisio C. 2013. Effects of the organic acids produced by a lactic acid bacterium in Apis mellifera colony development, Nosema ceranae control and fumagillin efficiency. Veterinary Microbiology 167(3–4): 474-83; doi:10.1016/j.vetmic.2013.07.030
- Moran, N. 2016. Genomics of the honey bee microbiome. Current Opinion in Insect Science 10: 22-8
- Mullen C, Frazier M, Frazier J, Ashcraft S, Simonds R, Vanengelsdorp D, Pettis J. 2010. High levels of miticides and agrochemicals in North American apiaries: implications for honey bee health. PLoS One 19;5(3): e9754
- Pătruică, S; Mot, D. 2012. The effect of using prebiotic and probiotic products on intestinal micro-flora of the honeybee (Apis mellifera carpatica). Bulletin of entomological research 102(6):619-23; doi:10.1017/S0007485312000144
- Raymann K, Shaffer Z, Moran N. 2017. Antibiotic exposure perturbs the gut microbiota and elevates mortality in honeybees. PLOS Biol 15(3): e2001861
- Samsel A, Seneff S. 2013. Glyphosate’s Suppresion of Cytochrome 9450 Enzymes and Amino Acid Biosynthesis by the Gut Microbiome: Pathways to Modern Diseases. Entropy 15(4): 1416-63; doi:3390/e15041416
- Servin, A. 2004. Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. FEMS Microbiology Reviews 28(4): 405–40; doi:10.1016/j.femsre.2004.01.003
- Shehata A, Schrödl W, Aldin A, Hafez H, Krüger M. 2013. The effect of glyphosate on potential pathogens and beneficial members of poultry microbiota in vitro. Current Microbiology 66(4):350-8
- Vásquez A, Forsgren E, Fries I, Paxton R, Flaberg E, Szekely L, Olofsson T. 2012. Symbionts as Major Modulators of Insect Health: Lactic Acid Bacteria and Honeybees. PLoS ONE 7(3): e33188; doi:10.1371/journal.pone.0033188
- Wu-Smart J, Spivak M. 2016. Sub-lethal effects of dietary neonicotinoid insecticide exposure on honey bee queen fecundity and colony development. Scientific Reports 6: 32108

Despite this common-sense logic appealing time and again to individual beekeepers, the message fails to reach beekeepers overall:
“Bees use bacteria to make pollen more palatable. The bacteria, mixed with pollen in beebread (the bees’ staple protein dinner), makes the pollen more easily digested. Herbicides kill bacteria. Obviously, if the herbicides kill bee-friendly pollen-reducing bacteria, herbicides may result in malnourished bees. This alone does not cause colony collapse disorder, nor has it alone caused the demise of bumblebees and other creatures. But it’s probably one more ingredient in the toxic soup that makes a bees’ life brief and dreary.” Ron Miksha
https://badbeekeepingblog.com/2016/04/28/herbicides-bacteria-killers-and-honey-bees/comment-page-1/#comment-6640