
Cold weather is here! Wrap your hives, snuggle in with a warm cup of coffee and venture into these interesting articles on a long November afternoon. In this digest: Overwintering in Canada. Understanding “crud brood”. Bee-bee recognition. Everything is connected. We can’t isolate a part of our planet from the rest of the ecosystem any more than various aspects of honey bee health can be isolated from honey bee gut microbes.
Dynamics of the Honeybee Gut Microbiota Throughout the Overwintering Period in Canada
Naomie Bleau, Sidki Bouslama, Pierre Giovenazzo, Nicolas Derome.
Food, Varroa, and Nosema all strongly influence the bee gut microbes. Old food increases Frischella perrara, a bacterium linked to bee gut abnormalities. Varroa infestation creates a dramatic change in honey bee gut microbiome, characterized by increased Snodgrassella alvi and decreased Lactobacillaceae in the gut of workers. And Nosema infection increases Gilliamella apicola.
Gut microbiota also varies considerably with time. For example, Gilliamella apicola dominates honey bee midgut in May, and Snodgrassella alvi becomes more abundant later in summer. Lactobacillaceae decrease later in summer as well. What happens with honeybee gut microbiota from October to May? Bleau et al. wonders: “Overwintering usually lasts from early December to April and is a critical period for the Canadian beekeeping industry. In the past 10 years, Canadian beekeepers have experienced large losses, with an average of 26% of their colonies dying while wintering or early in spring”. Bleau and colleagues at Université Laval followed nine honeybee colonies between September 2017 to June 2018 in north-eastern Canada to tease out the connections of food stores, Nosema, Varroa, and microbiology of the colony in winter.
“At the beginning of September, honey supers were removed, and colonies were reduced to one brood chamber. Fall feeding started in mid-September and all colonies were given 24 L of a sucrose 2:1 solution using a top box feeder (Wooden Miller feeder # FE-1100 from Propolis-etc., Beloeil, QC, Canada). Colonies received a Thymovar anti-varroa treatment starting on September 12, followed by an oxalic acid treatment on November 5 (drip method: 35 g/L in a sucrose 1:1 solution, 5 mL between every frame of the hive body crowded with honeybees). Colonies were wintered indoors in an environmentally controlled room (4–5 ◦C, 50–60% RH) from 22 November 2017 to 20 April 2018, and then moved to two spring apiaries until the end of June 2018. “
The researchers wrote: “We hypothesize that gut microbiota composition and diversity will follow a seasonal trend. We expect that microbial diversity will decrease in fall and winter because of antiparasitic treatments, isolation and the absence of plants to forage, which are an important source of microorganisms. Knowing how the microbial communities are affected by seasonal changes will allow us to identify gut dysbiosis in honeybees and propose probiotic nutritional supplements to improve the health of wintering colonies. “
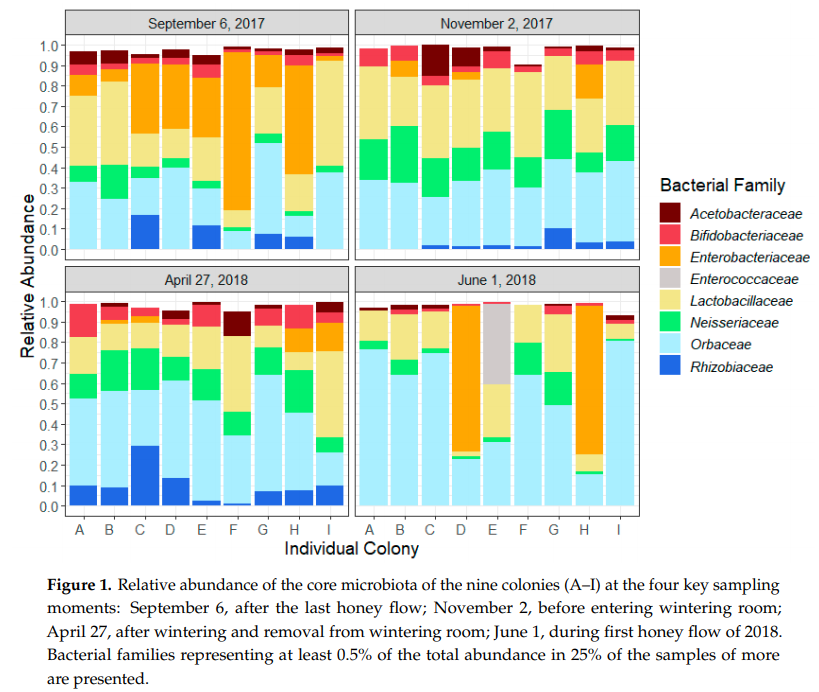
The data above shows microbial families in nine different colonies (A-I): Acetobacteriaceae (Bombella sp., Commensalibacter sp.), Bifidobacteriaceae, Enterobacteriaceae (Arsenophonus sp., Pantoea sp.), Lactobacillaceae, Neisseriaceae (Snodgrassella sp.), Orbaceae (Frischella sp., Gilliamella sp.) and Rhizobiaceae (Bartonella sp.).
September samples were dominated by the Enterobacteriaceae, shown by orange color. This family of bacteria is typically high in unhealthy colonies, and supports the researchers’ hypothesis that honeybee colonies experience dysbiosis in fall and winter. Authors note also: “Members of the Enterobacteriaceae family are among the main symbionts of Varroa destructor , and because of horizontal transfer between the mites and the honeybees [44], parasitized bees usually harbor higher levels of Enterobacteriaceae strains in their gut
Next, samples that had high abundance of unhealthy Enterobacteriaceae were deficient in beneficial Lactobacillaceae bacteria, shown in yellow.
Additionally, the more orange color there is in a given sample, the less yellow, green, and cyan it has. The only bacterial family that remained stable for the entire study was Acetobacteriaceae. Based on this study, there seems little need for Acetobacteriaceae-based probiotic, but seems likely a Lactobacilliaceae-based probiotic can improve the health of wintering colonies.
Amy S. Floyd, Brendon M. Mott, Patrick Maes, Duan C. Copeland, Quinn S. McFrederick, and Kirk E. Anderson
In 2016, a new honey bee probiotic candidate was announced: a strain called P.apium. Parasaccharibacter apium, later renamed Bombella apis according to more precise taxonomic identification, belongs to the Acetobacteriaceae family. P. apium project recruited many citizen scientists in a university-led effort to develop a new probiotic to protect honey bee from brood diseases, but the results indicate that hives fed this new probiotic did not get any benefits. Researchers from USDA-ARS, UA-Tucson, and UC-Riverside completed a new study on P. apium, showing that it has no survival benefit to healthy larvae, and no effect on European Foulbrood (EFB) infected larvae. This extensive study contradicts earlier results of 2014 study published by the same group of researchers. The difference was working out how to better manage honey bee larvae in the laboratory, very precarious at first. Authors write: “In our 2014 study… survival was exceedingly low… between the two trials at 40 and 70 percent survival”. Their new study increased survival of honeybee larvae to reliable 90% minimum. The earlier study showed that P. apium increased larval survival, but the later study revealed that it does not. Additionally, it revealed more than 30 strains of P. apium in larval guts, and only one strain may not yield a potent probiotic formulation.
While delivering a disappointing verdict on a potential of P. apium probiotic, this result is not very surprising given that P. apium (colored pink in the figure below)is already naturally abundant in honey bee food: stored pollen, honey, and in adult bees’ gut (this is also clear from the Canada overwintering study discussed earlier).
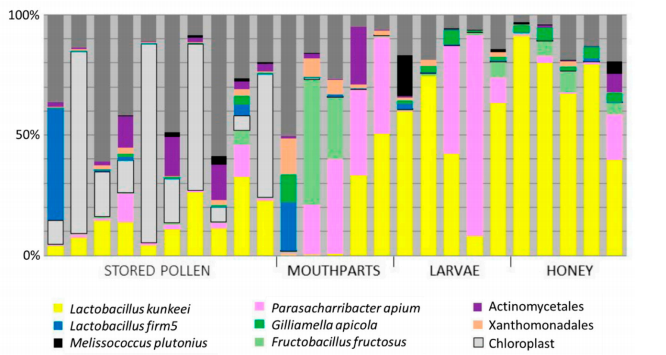
Importantly, the new study looked carefully at the survival of EFB-causing pathogen Melisococcus plutonius (colored black in the figure above) in different environments within the hive. The results indicate that “M. plutonius may be somewhat ubiquitous in disease free hives”. The results indicate that M.plutonius is relatively abundant in honey and 2nd instar larvae, while higher bacterial diversity of beebread and mouthparts is less likely to harbor M. plutonius. Thus, environments rich in non-native bacteria appear to play diminished role in EFB disease transmission. This finding is particularly important, since EFB and EFB-like disease are on the rise in North America and atypical EFB, also called “crud brood” causes significant economic losses to beekeepers; and antibiotic treatment prescribed to improve the condition has a risk of lowering bacterial diversity in the hive.
The gut microbiome defines social group membership in honey bee colonies.
Cassondra L. Vernier, Iris M. Chin, Boahemaa Adu-Oppong, Joshua J. Krupp, Joel Levine, Gautam Dantas, and Yehuda Ben-Shahar.
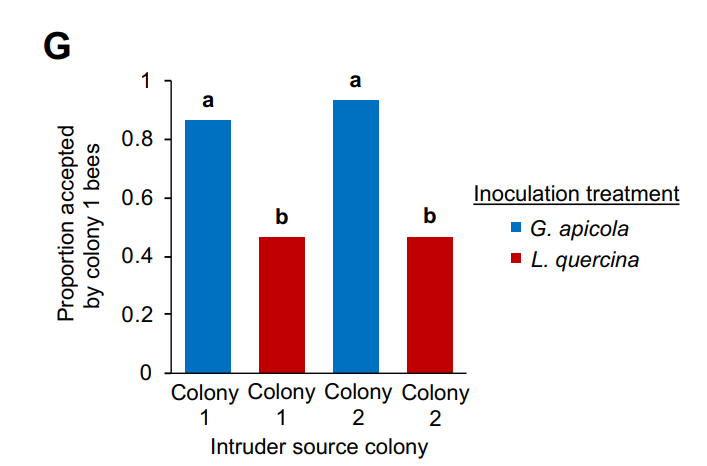
Researchers from Washington University showed that microbes in honey bee gut influence nestmate recognition behavior. Two factors identify honey bee as belonging to colony 1 or colony 2: colony-specific blends of cuticular hydrocarbons (CHCs) as pheromonal cues. These factors are acquired from nestmates, rather than controlled by bees’ genetic makeup. Experiments on sister bees treated with antibiotic and inoculated with different gut bacteria “indicate that acquiring different gut microbiomes, via social interactions with older bees, is sufficient to induce differences in CHC profiles between related individuals.” The bees that have different microbiomes and therefore, CHC profiles, are more likely to reject each other as intruders, while accepting bees with similar microbiomes.
It is unclear how closely these experiments reflect nestmate recognition in the field. However, microbial signature of adult bees may also play a role in rejecting those bees sickened or contaminated by pathogens: Lonsdalea quercina, one of the bacteria used to inoculate rejected bees, is a pathogenic bacteria present in drippy blight diseased red oak trees. Insects visiting diseased trees become contaminated and may be rejected.
Thank you for reading this science digest! Leave your question or comment below.

Excellent Work